 Lower orbital energies are correlated with lower (n+l) values.The (n+l) rule, which states that the orbital’s energy level is defined by the sum of its primary and azimuthal quantum numbers, can be used to identify the sequence in which the energy of orbitals grows.This suggests that electrons only move into orbitals with higher energy once orbitals with lower energies are fully occupied. The Aufbau principle states that electrons first inhabit orbitals with the lowest energies.Read Also: Henderson-Hasselbalch Equation Salient Features of the Aufbau Principle Additionally, the filling of electrons into orbitals within a single subshell must adhere to Hund’s rule, which states that before any two electrons pair up in an orbital, each orbital within a given subshell must be solely occupied by electrons. It’s crucial to remember that each orbital can only accommodate a maximum of two electrons (as per the Pauli exclusion principle). For instance, the electrical configuration of carbon is 1s22s22p2 and it possesses six electrons. The placement of electrons in an atom and the related energy levels can be understood using the Aufbau concept. Here, the terms “n” and “l” denote the principal quantum number and azimuthal quantum number, respectively. 
Germanic in origin, the word “aufbau” generally translates to “construct” or “build up.” Below is a schematic showing the sequence in which atomic orbitals are filled. The Aufbau principle states that atomic orbitals that are available and have the lowest energy levels are filled before those that have higher energy levels.  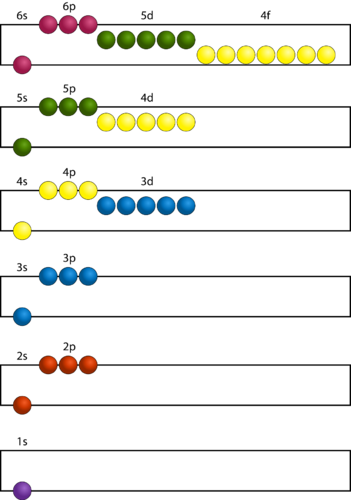
2.2 Writing the Electron Configuration of Nitrogen What is the Aufbau Principle?Īccording to this, electrons are inserted into atomic orbitals in ascending order of orbital energy. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
